Summary
The Pacific Toothed Oyster, Isognomon isognomon is a poorly studied pterioid in the past. However, in recent times, there has been a rapid increase of researches done in the understanding of this bivalve; due to its efficiency as a biomonitor. Unfortunately, despite this increase of studies and focus placed on this species, there was still a lack of studies done on the physiology and ecology of this species.
I.isognomon have inconsistent and irregular shapes due to its habituation in space-limited areas. Nevertheless, the inconsistency of shell shapes is similar throughout its genus Isognomon; with only a few small distinctive morphological characters used to differentiate species belonging to this genus. This species is also relatively young as compared to other bivalves; with its naming and taxonomy only being found in 1988 by Crampton. There is still lack of reproduction and ontogeny studies on this species, making it extremely difficult to fully understand this species of bivalve. I. isognomon also showed its ability to persist in a wide variety of regions, ranging from temperate to tropical. Its distribution has now been shown to be larger than what was previously thought; occurring only in northern Australia and the Indo-Pacific region.

External Morphology
External morphology.docx
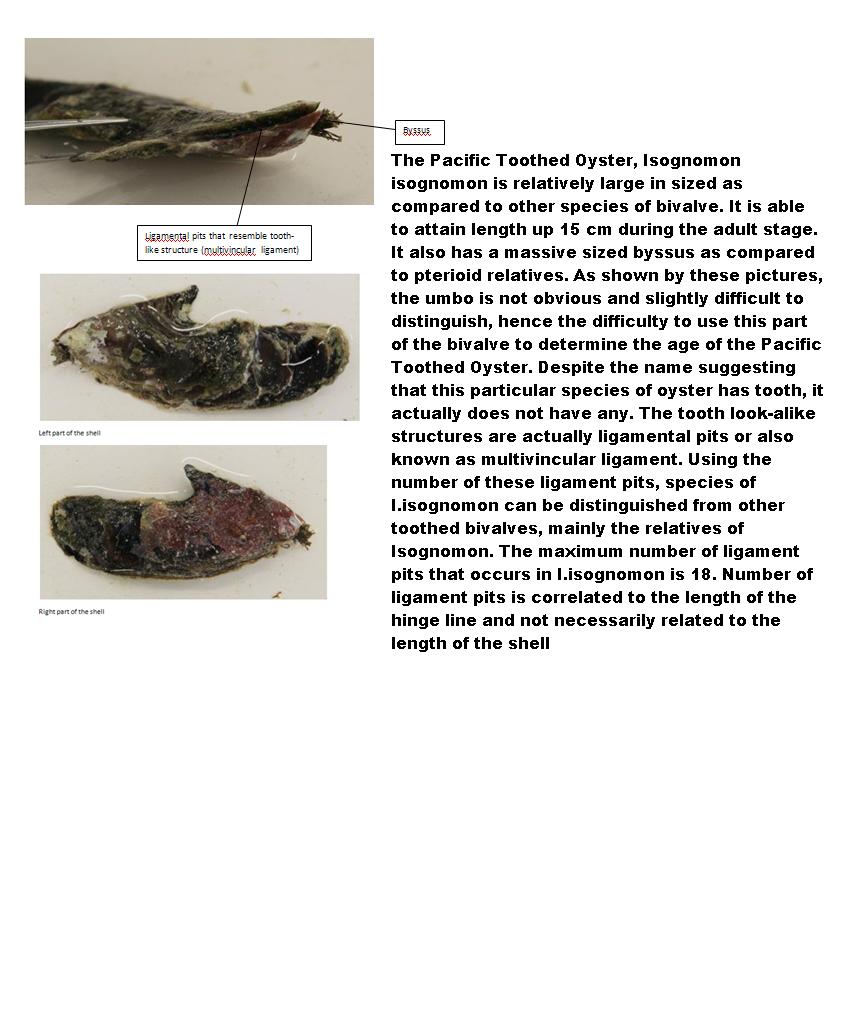 
Internal Anatomy of Pacific Toothed Oyster, Isognomon isognomon
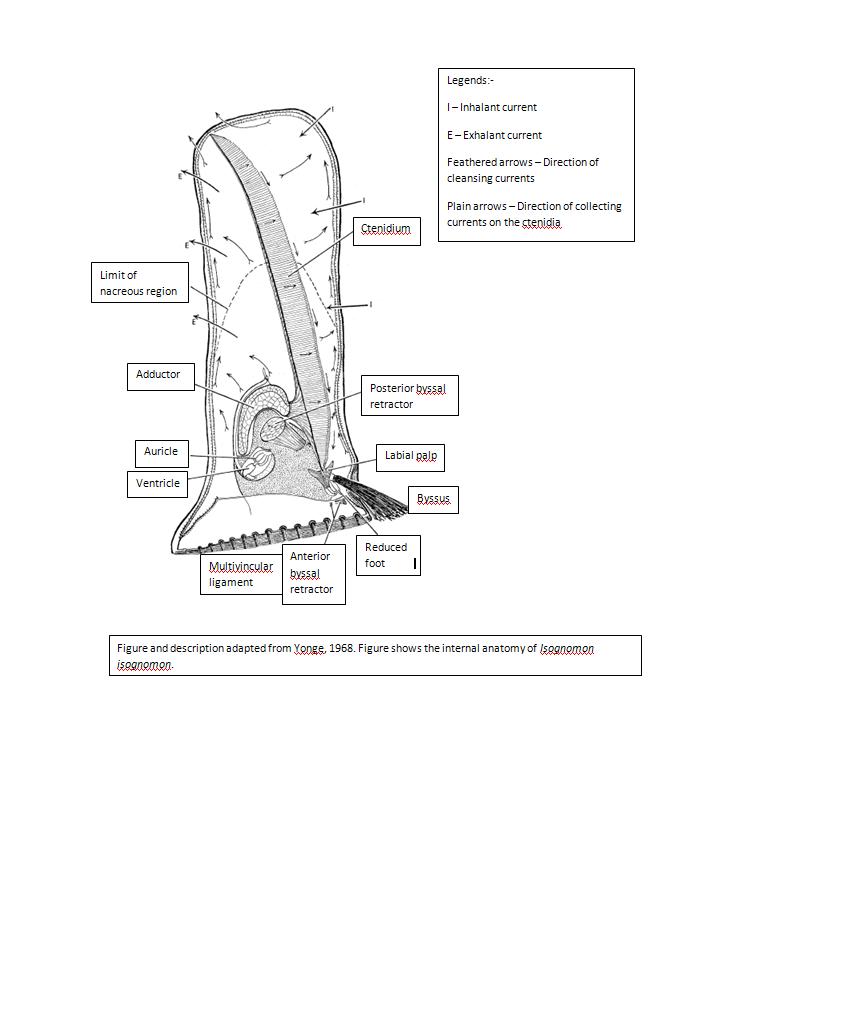  

Characteristics of Isognomon isognomon
Order Pteroimorpha
· Subclass of Bivalvia
· Large taxon of epibenthic bivalves
· Lamellibranch gills
· Attach to substrates using byssus or cementation
· Mantle margin not fused
· One foot is reduced
· Gills are usually large and assist in feeding
Superfamily Pterioidea
Family Isognomonidae
· Mesozoic species
· Tropical and warm temperate environment
· Typically equivalve or mytiliform in outline
· Umbones are incurved a little and some are not at all
· Lack of anterior auricle
· With or without anterior byssal gape, affecting both valves
· Commissure or joints are either planar or undulantes
· Surface ornaments are either smooth or concentrically lamellose. Some are all irregularly undulating
· Ligament areas are wide, flat with numerous narrow ligament pits
· Has single large, sub-centrally located adductor muscles
· Shell structure are relatively thick
Genus Isognomon
· Structure of the ligament extends along the entire dorsal surface of the shell
· Massive byssus
· Nacreous area is relatively large and its margins are less definite
· No pallial retractor
· No corresponding ride within distal region of the shell
· Straight hinge
· Toothless but has a series of short ligamental pits that resemble tooth-like structure
Species- Isognomon isognomon, Pacific Toothed Oyster
· Shells are straight or slightly curved (inconsistent)
· Hinge teeth is about equal to the width of shell
· Colouration of exterior- Inconsistent mixture of purple, black and brown
· Short posterior wing
· Large internal mother-of-pearl area but narrow non-mother-of pearl border
· Elongated and vertically disposed
· Can be found in clusters of single on the underside of rocky substrates and crevices
· Widely spreaded throughout Indo-Pacific region and northern Australia
 Global distribution of Isognomon isognomon Global distribution of Isognomon isognomon
The Pacific Toothed Oyster can be found widely throughout the Indo-Pacific region and northern Australia. However, through recent observations and occurrence data shown by Global Biodiversity Information Facility Network, the distribution of these crevice-dwelling pterioids is more widespread than previously thought. In addition to the previously thought distribution, Pacific Toothed Oyster can also be found around Eastern coastal areas.
(Yellow colour regions indicate distribution of Isognomon isognomon)
 ea of Australia, South East Asia, Southern Japan and Eastern Africa. ea of Australia, South East Asia, Southern Japan and Eastern Africa.
Types of Habitat
Pacific Toothed Oyster, Isognomon isognomon has an impressive ability to persist in a wide range of habitat. It is an epifaunal, benthic feeding species. With the exception of its dispersed lava, the adult form has no locomotive abilities. It can be found in water depths ranging from 0 to 3km depth of water. Its versatility to persist in different regions can also be expressed through its ability to persist in regions of subtropics, temperate and tropical conditions. Most of these bivalves can be found in marine waters but some were also found in estuarine and brackish environments (Yonge, 1968). The only definite habituation of these pteroids is that they are all attached to rocky substrates and in crevices. They are also relatively cryptic but have a high tendency to occur only on the underside of rocks and crevices (Tsubaki et al, 2010).
Overview of filtration rate experiment
Introduction
As there is an increase in demand of developing an effective biomonitoring system (Daby, 2006; Khristoforova et al, 2007; He´douin et al, 2006), there is a need in gaining a better understanding of the biological filtration rates of bivalves. Biomonitoring systems have been implemented to several parts of the world and is constantly used to assess the health of marine animals and water quality (Daby, 2006; Hédouin et al, 2010). The Pacific Toothed Oyster, Isognomon isognomon has been deemed to be among the more effective species to be used for the biomonitoring program (Daby, 2006; Hédouin et al, 2010). This is due to its ability to persist in a wide range of habitat, high abundance and high bioconcentration potential as well as having a history of successful utilization as part of the biomonitoring program (Daby, 2006). By using the biomonitors, there was a gained benefit by reducing the need to constantly collect water samples over a long period of time. Additionally, there would be lower rate of error gained as chemicals and heavy metals will build up in the body tissues of the bivalve (Hédouin et al, 2010; Daby, 2006).
Despite all the benefit gained, there was still a need to sacrifice individuals to gain tissue samples for data collection. Though this species is not endangered in any manner, given a choice to decrease sacrifices for data collection would always be preferred. On top of that, there were no experiments to show the differences of results accuracy between conventional methods of human performed data collection against biomonitoring methods. In this experiment, a simple filtration rate will be tested to gain a better understanding of the percentage materials in water that will be enveloped by these bivalves over a period of time. On top of that, it will also allow us to gain a better insight of how long will it be needed for these bivalves to filter materials in the water as well as learning if turbidity of water can be altered given the right amount of individuals. For this experiment, it was hypothesized that water turbidity will keep decreasing over a period of time as materials in water will be filtered and retained in the tissues of the bivalves.
Methods
In this experiment, 15 individual subjects were placed in a container holding 15 liters of filtered sea water. After placing the individuals in, water samples were taken straight away to be used as a control. After collecting the control water sample, 6 ml of microalgae solution was added and another water sample was taken. Consequently, water samples were taken up to 11 times. The first 7 water samples were taken at every one hour interval after the experiment was set up; whereas the following water samples were taken 11 hours later. Finally, 3 water samples were collected at each hour interval following the previous collection of water samples. Every collection of water samples were placed in test tubes and kept in a -4°C environment to prevent any build up unwanted material. The entire set up experiment was left in an open environment which experienced temperatures between 20-24 °C to simulate a natural occurring environment. Once data collection was done, all water samples were placed in a spectrophotometer to gain data collection with regards to the percentage of water turbidity.
Results & Discussion
The results of the experiment was confounding, with unexplained variables detected during the data analysis. The result are as shown:-
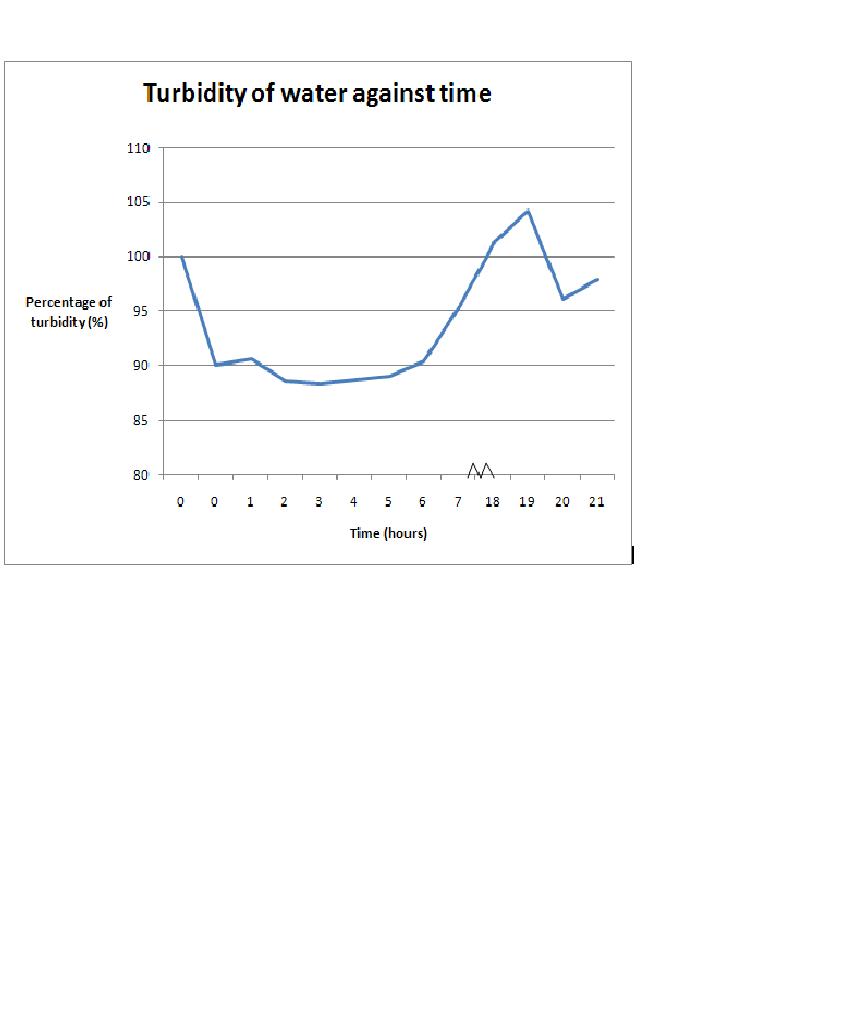 
Graph showing the variability of turbidity percentage in the water over a period of time. First 2 water samples were controls used for comparison purposes. The first water sample is filtered sea water (expected to be clearest) and second water sample consists of 6ml of microalgae solution (expected to be the most turbid). Eventual samples are for data analysis purposes over the duration of time.
As the results show, the initial hypothesis was rejected. For the first 6 hours, water became more turbid than expected. Between the 7th hour to the 19th hour, turbidity of water started to decrease; however, it was unexpectedly clearer than the first water sample that was thought to be the clearest. Additionally, after the 19th hour, there were unexpected fluctuations again. It was shown in this result that the fluctuation of water turbidity is extreme and results were confounding.
As the results suggest, collection of water sample over a period of time for monitoring marine water cleanliness may not be highly recommended as there were thought to have high variables that are too stochastic to be taken into account. Using this experiment for an example, on the surface of the bivalves, there were communities of invertebrates and algae that settled on it. These communities may have altered the result of this experiment; however, further investigation will be needed to determine such hypothesis. Additionally, there were no previous reports over this event. A further understanding and learning of these settled communities on the shell surface may be important for future implication as biomonitors as there may be symbiotic or parasitic possibilities that may affect its effectiveness to filter materials. Another unknown variable in this experiment is if the filtered water that leaves that cavity of the bivalves are meant to be clearer or does it carry other form of substances from the body such as excretion. Similar to the first case scenario, there was no concrete experiment or research that shows such possibilities. Understanding of the physiology and ecology of the Pacific Toothed Oyster can be vital for future research and understanding. At this current stage, this species of bivalves are much understudied other than for biomonitoring purposes. The physiology and ecology that occurs on and around this species is very poorly understood and may well be a limitation for future implication as biomonitors.
Reproduction and Life Cycle
Little is known about the reproduction and life cycle of the species in the Isognomonidae family. The prodissoconchm, larval shell of isognomonids is similar to a planktotrophic pterioid. This prodissoconch suggests the possibility of species in the family of Isognomonidae to be of broadcast spawners of their larva. The dispersed larva is also thought to undergo lecithotrophic development. The umbones are usually used as references of the earliest ontogeny of bivalves, however for these species belonging to the Isognomonidae family, this is not true. The umbone is not obviously gyrate and thus inhibiting possibility of using the umbone as reference of it ontogeny. Additionally, the larval shell and ligament growth are also poorly studied in the past and yet to be concluded. (Malchus, 2004)
Fossil History
History of naming and fossil discovery
Initially in 1976, the first specimen of the pacific toothed oyster was misidentified and thought to be part of the Inoceramidae family. In 1988, Crampton reassessed this species of bivalve and clarified that it represents a species of Isognomon. Following this assessment, Crampton gave it a new name which is also currently used to identify this species. The scientific name that was given is Isognomon isognomon. I. isognomon was also clarified to be of separate as compared to other bivalves that occurs during the Late Cretaceous and Palaeocene period. The distinctive characters that distinguish Isognomon sp. against bivalves that occurred during Late Cretaceous and Palaeocene period are delicate resilifers and larger size shells.
(Stillwell, 2010; Crampton, 1988)
 
Morphology Differences Between Sister Taxa
Classification of the pacific toothed oyster can be considered to be difficult due to the irregularity of shell shape size. Additionally, sister species in the genus Isognomon also do have high amount of irregularity and inconsistency in the shell structure, ultimately making classification of these species just by morphology to be difficult.
Pacific toothed oysters, Isognomon isognomon have high variation of shell structure among individuals despite classified as being the same species. This high variation was hypothesized to be driven by ecological and physiological pressure such as predation and water quality. However, after further investigation over this matter, it was shown that the shell morphology variation is high even among species belonging to Isognomidae family. Crevice-dwelling isognomons are bivalves that attached themselves to the underside of rocky substrates in shallow waters (Kauffman, 1969). As these habitats restrict the amount of space available, these bivalves are forced to alter their final adult size and shape accordingly, resulting from distorted shell shapes (Ubukata, 2003; Tsubaki et al, 2010).
A study by Ubukata in 2003, demonstrated that the variety of shell shape of the Pterioida family have sets of common prescribed directions. These set of prescribed directions are strongly driven by types of habitat that the individuals are grown in. Shell structure are highly based on the speed and energy of the flow of water current, salinity of the water, predation exposure, types of attached substrates and the amount of materials present for calcification (Ozanna et al, 2002; Ubukata, 2003). Additionally, the growth and shaping of the shells are also highly restricted by ontogenetic growth rules. Due to the ontogenetic rules, restrictions were applied during morphogenesis, causing restrictions on direction of shell margin extension and the direction of mantle extension. Each species has its own rate of ‘polarity’, which causes morphological differences between species in a latter growth stage (Wilson, 1993; Ubukata, 2003).
Applied Biology (Biomonitor of water quality)
Most economically important cities are located near to coastlines. With this said, these cities play a major role in contamination and disruption of the marine ecosystem. Marine ecosystems have been clarified to have a very dynamic environment and ecosystem; nevertheless, they are fragile to anthropogenic activities (Daby, 2006; Hédouin et al, 2010). Additionally, there are also non-city regions that are undergoing pollution pressures due to mining and dredging activities (Brown & Kumar, 1990; He´douin et al, 2006; Khristoforova et al, 2007). Human-induced activities causes a runoff of chemicals and heavy metals such as Cromium, Lead, Zinc, Cadmium, Iron, Nickel, Magnesium and Copper (Daby, 2006; Khristoforova et al, 2007; He´douin et al, 2006; Hédouin et al, 2010). These chemicals and heavy metals are non-biodegradable and have a long lasting effect in the marine environment (Hédouin et al, 2010). As this runoff of heavy metals is highly toxic to the marine environment, there was and there still is a strong demand for a monitoring scheme or system to be placed upon critically affected areas (Daby, 2006; Khristoforova et al, 2007; He´douin et al, 2006).
With the urgency of providing a monitoring program, clean and effective monitoring management were developed using bivalves. Biomonitoring approaches are advantageous as compared to direct measurements of water quality using conventional methods, which is a time-integrated assessment (Hédouin et al, 2010). Using biomonitoring methods, a long term and effective build up of chemicals can be detected better as compared to conventional methods of collecting water samples for an extensive period of time and then running laboratory test on them.
There were a couple of marine invertebrates that were previously used for the biomonitoring program; however, bivalves were deemed to be a better choice due to lack of motility, high filtration rate of water and easily accessible accumulation of chemicals and metals in body parts (Brown & Kumar, 1990; Daby, 2006; Khristoforova et al, 2007).
The Pacific Toothed Oyster, Isognomon isognomon was identificed to be among the more successful species of bivalves to be used as a part of the biomonitoring program (Daby, 2006; Hédouin et al, 2010). This was due to its common occurrence, having a high bioconcentration potential and as well as portraying successful utilization in the past (Hédouin et al, 2010; Daby, 2006). Additionally, in recent findings by Global Biodiversity Information Facility Network, this species has an ability to persist in a wider range of environment than previously expected, making this species as an even more attractive choice for biomonitoring programs.
References
Kauffman, E.G. (1969). Functional morphology of bivalve shell. Treatise on invertebrates paleontology, part N, Mollusca 6, Bivalvia Boulder,Geological Society of America & University of Kansas, N410-n183
Tsubaki, R., Kameda, Y., & Kato, M. (2010). Pattern and process of diversification in an ecologically diverse epifaunal bivalve group Pterioidea (Pteriomorphia, Bivalvia). Molecular Phylogenetic and Evolution 58: 97-104
Ubukata, T. (2003). A morphometric study on morphological plasticity of shell form in crevice-dwelling Pterioida (Bivalvia). Biological Journal of the Linnean Society 79: 285-297
Yonge, C.M. (1968). Form and habit in species of Malleus (Including the “Hammer Oysters”) with comparative observations on Isognomon isognomon. Biological Bulletin 135(2): 378-405
Ozanna, C.R., & Harries, P.J. (2002). Role of predation and parasitism in the extinction of inoceramid bivalves: an evaluation. Lethaia 35: 1-19
Savazze, E. (1995). Parasite-induced teratologies in Pliocene bivalves Isognomon maxillatus. Palaeogeography, Palaeoclimatogy, Palaeoecology 116: 131-139
Khristoforova, N.K., Kavun, V.Y., Latypov, V.Y., Tien, D.D., Zhuravel, E.V., & Tuyan, N.Y. (2007). Heavy metals in mass species of bivalves in Ha Long Bay (South China Sea, Vietnam). Oceanology 47(5): 685-690
He´douin, L., Pringault, O., Metian, M., Bustamante, P., & Warnau, M. (2007). Nickel bioaccumulation in bivalves from the New Caledonia lagoon: Seawater and food exposure. Chemosphere 66: 1449-1457
Daby, D. (2006). Coastal pollution and potential biomonitors of metal in Mauritius. Water, Air and Soil Pollution 174:63-91
He´douin, L., Metian, M., Lacoue-Labarthe, T., Fichez, R., Teyssie, J-L., Bustamante, P., & Warnau, M. (2010). Influence of food on the assimilation of selected metals in tropical bivalves from the New Caledonian lagoon: Quanlitative and quantitative aspects. Marine Pollution Bulletin 61: 568-575
Brown, B.E., & Kuman, A.J. (1990). Temporal and spatial variations in iron concentrations of tropical bivalves during a dredging event. Marine Pollution Bulletin 21(3): 118-123
He´douin, L., Metian, M., Teyssie, J-L., Fichez, R., & Warnau, M. (2010). Delineation of heavy metal contamination pathways (seawater, food and sediment) in tropical oysters from New Caledonia using radiotracer techniques. Marine Pollution Bulletin 61: 542-553
Stilwell, J.D. (1998). Late Cretaceous Mollusca from the Chatham Islands, New Zealand, Alcheringa. An Australasian Journal of Palaeontology 22(1): 29-85
Malchus, N. (2004). Constraints in the ligament ontogeny and evolution of Pteriomorphian Bivalvia. Palaeontology 47(6): 1539-1574
Crampton, J.S. (1988). Comparative taxonomy of the bivalve families Isognomonidae, Inoceramidae, and Retroceramidae. Palaeontology 31(4): 965-996
Wilson, B., Wilson, C., & Baker, P. (1993). Australian marine shells. Odyssey Publishing. Leederville, W.A.
Stang, D. (2004). Isognomon isognomon. Available: http://www.poppe-images.com/photo/collection/970000/web/974431.jpg. Last accessed 24th October 2011.
Isognomon (Isognomon). The Paleobiology Database. Available: http://paleodb.org/cgi-bin/bridge.pl?a=checkTaxonInfo&taxon_no=97432&is_real_user=1. Last accessed 23rd October 2011.
Global Information Biodiversity Facility. Available: http://data.gbif.org/species/14782199. Last accessed 22nd October
Taxonomy
Domain: Eukaryota – Whittaker & Margulis, 1978
Kingdom: Animalia- Linnaeus, 1758
Subkingdom: Bilateria – Hatschek, 1888; Cavalier-Smith, 1983
Infrakingdom: Lophotrochozoa
Phylum: Mollusca- Linnaeus, 1758; Cuvier, 1795
Class: Bivalvia – Linnaeus, 1758
Subclass: Metabranchia
Superorder: Filibranchia
Order: Pteriomorpha/ Pterioida- Newell, 1965
Suborder: Pteriina – Newell, 1965
Superfamily: Pterioidea – Gray, 1847
Family: Isognomonidae- Woodring, 1925
Genus: Isognomon – Solander in Lightfoot, 1786
Scientific name- Isognomon isognomon – Crampton, 1988
Sister taxa
Sister taxa (members belonging to the genus Isognomon)
· I. acutirostris
· I. alata
· I. alata bicolor
· I. alatus
· I. albisoror
· I. anomioides
· I. attenuate
· I. bathonicus
· I. bicolor
· I. biplicatum
· I. bouchardi
· I. californicum
· I. chemnitzianus
· I. clarki
· I. costellatus
· I. ephippium
· I. flambarti
· I. flamberti
· I. hataii
· I.incisum
· I.isognomoides
· I. isognomonoides
· I. isognomum
· I. isogonum
· I. janus
· I. janus orientalis
· I. oxoniensis
· I. perna
· I. pernum
· I. promytiloidea
· I. radiate
· I. radiates
· I. recognitus
· I.rectangularis
· I. ricordeanus
· I. rollei
· I. rugosus
· I. rupella
· I. semicurita
· I. subplanum
· I. sulcata
· I. thurmanni
|