Microplastic Study
Ingestion and Translocation of Microplastics in the Ecteinascidia diaphanis
Introduction
Global plastic production has increased dramatically in recent years, from 5 million tonnes in the 1950s to over 280 million tonnes in 2011 (Plastics Europe, 2012). Plastic has a high durability and along with this increase in production there is a widespread accumulation of discarded plastic in all habitats, including aquatic areas (Derraik, 2002). Plastic is known to progressively break down into smaller pieces, fragments of plastic less than 5mm are said to be microplastics, although this is not yet universally classified (Cole, 2011). The effects of large pieces of plastics upon marine wildlife have been well documented; these include ingestion by over 180 species leading to starvation and suffocation (Derraik, 2002). However, until recently the impact microplastics may be having on our oceans was not recognised.
Microplastics occupy the same size range as sand grains and planktonic organisms, meaning it is likely to be ingested by a wide range of invertebrate filter feeders, near the base of the food chain. It has been previously shown that amphipods, barnacles and lugworms can ingest microplastics (Thompson et al., 2004). Once microplastic is ingested there are many pathways it could take; it may be retained in the digestive tract, be egested through defecation, or transferred through the epithelial lining of the gut into body tissues (translocation) (Browne et al., 2008). Browne et al. (2008), suggested that ingested particles of microplastic can translocate from the gut cavity to the circulatory system of mussels, and smaller particles were translocated more readily than larger particles.
I chose to look at the effects microplastics may have upon E. diaphanis. Ascidians are found in great abundance with the second largest biomass to porifera in their cryptic communities. They are incredibly efficient filter-feeders (see Digestive System, Nutrition and Excretion) and are therefore responsible for removing particles from the water column. They are known to take up and store toxic substances, purifying the surrounding water (Ruppert et al., 2004), making them important invertebrates within their ecosystem. Ascidians contribute to the marine food web as a low level species preyed upon by specialised molluscs, echinoderms and fish (Stoeker, 1980; Parry, 1984).
With an increase in microplastic abundance it would be expected that ascidians would be affected, in turn affecting the ecosystem and leading to accumulations within higher trophic levels. As with all animals it would be expected that microplastic ingestion would have negative impacts, whether this is through starvation or translocation affecting tissues and other mechanisms. The species E. diaphanis was chosen due to its lack of pigmentation allowing the uptake and passage of microplastics to be seen.
Methods
This study was carried out on Heron Island (23.441947 S,151.91268 E), Great Barrier Reef, Australia. Heron Island is a lagoonal reef island housing a great variety of species. A number of E. diaphanis were collected from boulders on the North beach, Heron Island. Theses zooids were immediately transported back to the field laboratory in a plastic container. Individuals were stored collectively in an open container within a constantly aerated aquarium, which used a flow though seawater system, pumped directly from the sea. Upon completion of the study all zooids were returned to the ocean.

|
|
Figure 1: Photograph showing the differently sized microplastic beads used in the study. |
Three zooids were placed in a container of approximately 10cm in diameter, containing 300ml solution of seawater obtained from the aquaria flow through system and plastic spheromeres (beads). The spheromeres used were 10µm, 20µm and 40µm. A control was also used which contained 300ml of seawater from the same flow through system. Ten containers were set up (inclusive of the control), each containing a different solution. The solutions used were 50, 000 beads per ml, 250, 000 beads per ml and 500, 000 beads per ml. A hemacytometer was used to calculated and verify each solution. Each solution was tested with each bead size.
The zooids were subjected to the treatments for two hours, using a magnetic stirrer to keep the beads in suspension. After two hours the zooids were placed in small containers with fresh seawater. Photographs were then taken of the zooids immediately, 2 hours after and where possible 24 hours after treatment. These were taken using a microscope eyepiece camera at a magnification of 0.8. The heart diameter was then measured from the photographs and compared to the control.
Results
All zooids ingested the beads and could be seen in the oesophagus. The amount of plastic translocated into the circulatory system varied with bead size and concentration.
  
Figure 2: Images displaying zooid hearts immediately after being removed from the microplastic solution. From left: 50,000 concentration of 10µm bead; 250,000 concentration of 40µm beads; 500,000 concentration of 20µm beads.
As the concentration of plastic increased so did the heart diameter. There was a significant increase in the heart diameter between the 500,000 and 250,000 concentrations and the control.
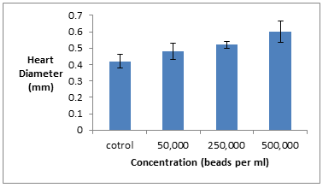
Figure 3: Chart showing average zooid heart diameter for each concentration of solution tested. Error bars denote standard deviation found in heart diameter.
The greatest increase in heart diameter was found in those subjected to 20µm beads. There was a significant difference in the heart size of the control group and the 20µm and 40µm groups.
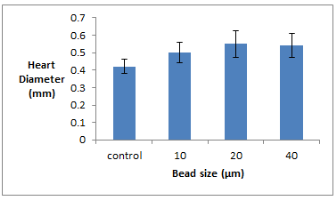
Figure 4: Chart showing average zooid heart diameter for each microplastic bead size tested. Error bars denote standard deviation found in heart diameter.
From observations it was also noted that the lower the concentration and the smaller the beads, the shorter the time taken to remove the plastic from the body.
Discussion

|
Figure 5: Photographs of zooids after being in microplastic solution. Microplastic beads can be seen in the pharyngeal basket.
Left: concentration 500,000 size 10µm. Right: concentration 500,000 size 20µm. |
All sizes of microplastic beads at all concentrations were ingested and transported to the gut, where they accumulated in the digestive tract, but were egested in the faeces or purged from the pharyngeal basket. It was also seen that the beads translocated from the gut cavity to the circulatory system in a very short time, less than two hours. The majority of previous research investigating the ingestion of macroplastics in marine invertebrates has only shown that it may be retained in the digestive tract (Derraik, 2002) or egested in the form of faeces (Eriksson and Burton, 2003). This study shows that these are not the only ways in which microplastics can travel through the body. From my observations I am not able to determine the mechanisms in which the beads were translocated, further work is needed to determine the mechanisms by which these particles are taken up across the gut membranes and accumulate in the blood.
The presence of microplastic beads within the circulatory system could restrict blood flow causing damage to the vascular tissues and slight changes in cardiac activity could be seen. In my study there was an accumulation of beads in the circulatory system for all sizes and concentrations, it therefore seems likely that both sizes translocate by the same mechanisms. Browne et al. (2008), suggested that the translocation of microplastics from the gut to the hemolymph in mussels may be a result of phagocytosis, it is possible that this is also the case in ascidians. However, they suggest that if phagocytosis is involved, then smaller particles are more easily phagocytosed, possibly because the phagosomes within each cell can accommodate a greater number of smaller particles. This was not seen in my study, I found that the mid-size beads were the most readily translocated, with the smallest accounting for the least difference in heart diameter. This may be due to the smallest of my beads being the same size as a cell, therefore may pass through membrane readily and may not increase the volume of fluid as much as larger particles. Further research is required to test these predictions and to determine the upper and lower size boundaries for ingestion and translocation of plastic debris in other organisms.
The microplastics were removed from the body in a very short time, either via faecal egestion or purging. No adverse effects could be seen in any of the zooids as a result of microplastic ingestion. This was unexpected due to the high concentrations the zooids were subjected to. However, it is premature to suggest that ingestion and/ or translocation of microplastics does not cause any long-term or toxicological effects. My study exposed E. diahpanis to the microplastic solutions for 2 hours and where possible were monitored for a very short time period, 24 hours at most. In their natural environment organisms will be subject to microplastics over much longer time scales, and the effects of this remain unclear. Various types of plastics are known to contain and release toxins. Ascidians are accumulate toxins from the water column, which are then stored until the death of the zooid (Ruppert et al., 2004). It is possible that the zooids are accumulating toxins released from the microplastics. Further research is needed to examine the toxicological consequences of long term exposure to various microplastics in marine habitats. Particularly, as it has now been shown that some invertebrates translocate microplastic into their circulatory system, allowing any associated toxins to be distributed throughout the body.
The ingestion of microplastics by E. diaphanis would not appear to have short term effects upon the zooids. It would appear that the zooids of this species are able to ingest and egest or purge microplastics at no apparent cost to themselves, however, long-term effects were not studied and remain unkown. As these zooids were subjected to unreasonably high concentrations of microplastics, it is likely that microplastics in their natural environment will not pose a threat to them. It is unclear as to whether due to their ingestion microplastics will accumulate and have adverse effects in higher trophic level species. |