Anatomy & Physiology
One specimen of M. rosea was discovered on a small fragment of coral rubble collected from the Fourth Point reef at Heron Island. Over the course of a week, this species was kept under observation at the Heron Island Research Station to investigate its behaviour. After noting behavioural observations the small specimen (6mm) was relaxed by exposing it to -4oC for approximately five minutes, followed by fixing the animal with Magnesium Chloride and allowing twenty-four hours for the fixative process to complete. Once fixed, the specimen was preserved in 70% ethanol and used for sectioning with H and E staining.
A research project was developed for the specimen of M. rosea surrounding the internal anatomy and physiology to determine the defence mechanism in this species. Minimal research on this particular nudibranch and confusion surrounding its taxonomic place in a suitable family, has led researchers to question its method of defence. Does M. rosea utilise the cnidocytes from its hydroid prey, or does this nudibranch employ a chemical defence mechanism to warn off predators?
The cerata of this animal were dissected and mounted onto slides to observe the internal morphology. The remaining body of the nudibranch was sectioned and stained with H and E dye. Through the process of sectioning and staining, this project aimed to clarify the defence mechanism of M. rosea and investigate the internal anatomy and physiology of the animal, in order to provide extensive knowledge on this uncommon species.
FEEDING AND DIGESTIVE SYSTEM
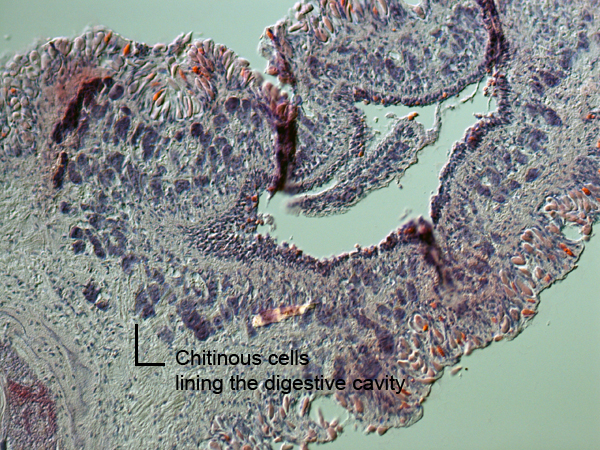
The presence of a large darkened shadow inside the animals body, suggests that the animal had recently eaten and was undergoing digestion when the specimen was first located. The digestive cavity can be observed in the image below. The digestive tract of M. rosea is lined by thick chitinous cells (dark blue/purple spots), which aid in structural support of the organelle. Cuticularization within the stomach is present in M. rosea, a common anatomical feature of the Tritoniids (Willan 1988).
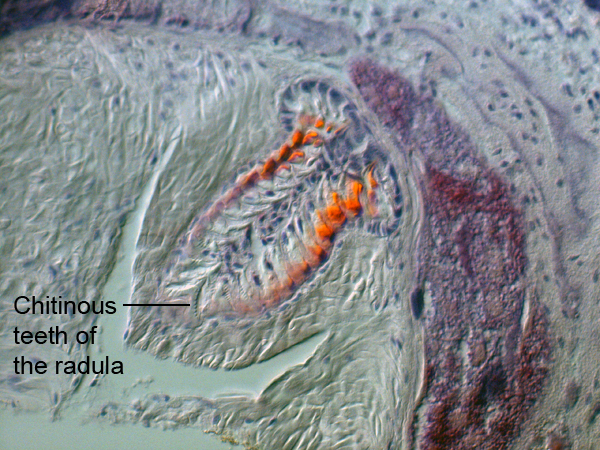
The radula is a unique feeding apparatus of all Molluscan species. In M. rosea, the radula is used to scrape hydroid material into the buccal cavity, that is then passed into the oesophagous and further into the digestive cavity. In this species, the radula comprises six lateral rows of chitinous teeth that more closely resemble the structural morphology of an Aeolididae (Willan 1988). These chitinous teeth can be observed in the following image of the radula. Most teeth have denticles (small tooth-like projections), which is also characteristic to the morphology of Aeolid nudibranchs (Willan 1988). The posterior end of the radula is reinforced with minerals, the pink and orange highlighted sections, that aid in structural support and movement of this specialised feeding apparatus.
The buccal cavity is considerably large in proportion to the rest of the animal’s body, and encloses the radula within it. Tubercles are present on the jaw’s masticatory border. These are generally sites of muscle insertion that aid in movement of the jaw (Willan 1988). The surrounding buccal cavity is also highly muscularised to support movement of the mouth during feeding. Salivary glands are found to be surrounding the buccal cavity, which are known to secrete mucous to aid in digestion when the animal acquires a food source (Behrens 2005). The oesophagous extends directly from the buccal bulb into the stomach. What is unique to this species is that it lacks an oral veil, which is often observed in species of 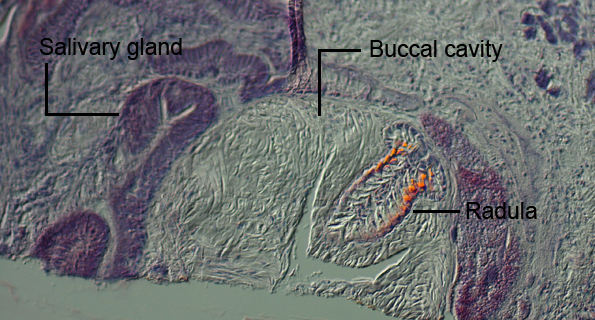 Tritoniids. The lack of this anatomical feature resembles more closely the body plan of an Aeolididae. Tritoniids. The lack of this anatomical feature resembles more closely the body plan of an Aeolididae.

MUSCULATURE AND DEFENCE
The small body plan of M. rosea means that this species does not require high internal muscularization. Muscle is present to enable the animal to engage in behavioural activity of swimming and rearing.
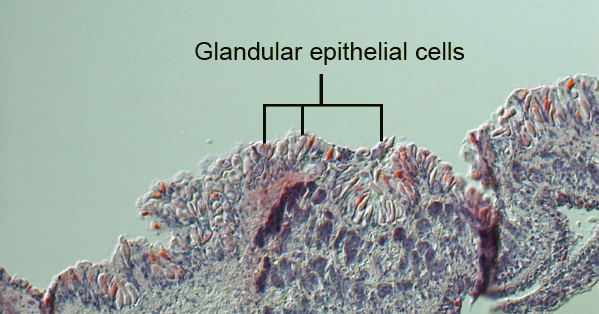
Members belonging to the Tritoniidae family have defensive subepidermal chitinous granules or glandular cells that cover their body (Wagele et al. 2006; Martin et al. 2007).These granules are prominent in the oral tentacles, rhinophores, and cerata. As nudibranchs have evolved to survive without the use of a chitinous shell, these animals have utilized the protein in the epithelia of their body plans as a protective mechanism. The cells are utilized by other species of Tritoniids to encapsulate cnidocytes that are ingested from the prey (Martin et al. 2007). Their role is to neutralize the effect of toxins from the cnidocytes that are delivered to the nudibranch. In most Tritoniids, diverticulum from the digestive glands does not penetrate the cerata, which is how cnidocytes are effectively transported to ceras tips in Aeolid species. The specimen of M. rosea discovered on Heron Island was far too small and delicate for cerata sections to be informative. Alternatively, cerata from the mid-right dorsal section of the animal was mounted and observed for the presence of cnidosacs containing cnidocytes. The image of this cerata shows no presence of cnidosacs or cnidocytes, suggesting that this species does not utilise the cnidocytes from its hydroid prey.
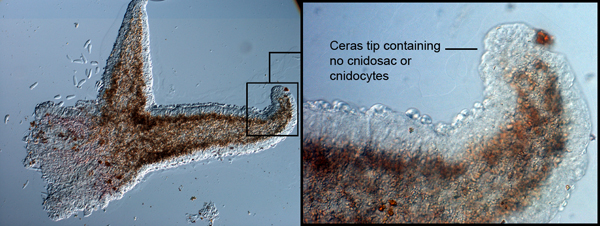
The absence of cnidosacs and cnidocytes in M. rosea confirms that diverticulum from the digestive glands does not extend into the cerata of this species. The absence of a mechanical defence mechanism suggests that Marianina rosea may employ the use of subepidermal chitinous granules or glandular cells to diffuse the effects of cnidocytes from its hydroid prey and engage in a chemical defence mechanism. Extensive research into this method of defence would provide further understanding of the exact defence mechanism employed by M. rosea. The defensive glandular cells that aid in protection can also be observed in the epithelia of the body of M. rosea.
REPRODUCTIVE SYSTEM
As it was unclear as to whether the specimen located on Heron Island was a sexually mature adult, sections did not provide significant detail on the reproductive aperture of this species. It has been suggested, however, that the reproductive organs of M. rosea reflect the arrangement and structure of organs found in other Trioniid nudibranchs. The reproductive organs are located on the right side of the animal’s body, roughly a third of the way down the back in between the rhinophores and the first pair of cerata. In M. rosea, the distal hermaphrodite duct is separated into the vas deferens and the oviduct, which are both located within the albumen gland (Willan 1988). The ovotestis are arranged around the posterior section of the left digestive gland. This particular species of nudibranch lacks a distinct prostate gland (Willan 1988).
SENSORY SYSTEM
Palmate rhinophores are the defining feature of M. rosea. These morphological structures are incredibly sensitive, allowing the animal to detect suitable food sources, predators and potential mates over great distances. The sensitivity of the rhinophores is due to the presence of olfactory glomeruli, that aid the animal in processing important chemical information of the surrounding aquatic environment (Werts et al. 2006). These glomeruli are observed in the rhinophoral base as randomly distributed pink circular masses.
The base of an oral tentacle can also be seen in the image below. The oral tentacles are utilised by the nudibranch for touch, allowing the animal to feel its way around substrates in search of food.
The anatomical and physiological study performed on Mariana rosea, located on Heron Island, has confirmed that this species of nudibranch does not employ a mechanical defence mechanism. The exact defence mechanism of this species is still unclear with further research warranted on the ecology, and anatomy and physiology of this uncommon nudibranch.
|